X-rays have existed since Roentgen’s discovery of them in 1895. They have been used for a variety of purposes, such as diagnosing health conditions in a doctor’s office and detecting prohibited objects at an airport.
X-rays can pass through objects, which absorb varying quantities of radiation depending on their composition. This allows us to create images of objects such as fractured bones.
Now, a team of scientists from the University of Ohio and Argonne National Laboratory directed by Professor Saw Wai Hla has conducted new research.
They were capable of capturing an X-ray of a single atom. This is significant because it may have a significant impact on materials science and possibly even medicine.
The scientists detailed their accomplishments in the scientific journal Nature.
Utilization of ‘Synchrotron X-ray’
The scientists utilized synchrotron X-ray technology, a specialized type of X-ray technology. This involves accelerating electrons to produce an extremely potent source of energy.
Prior to this research, it was challenging to photograph anything smaller than 10,000 atoms. However, the team discovered a solution.
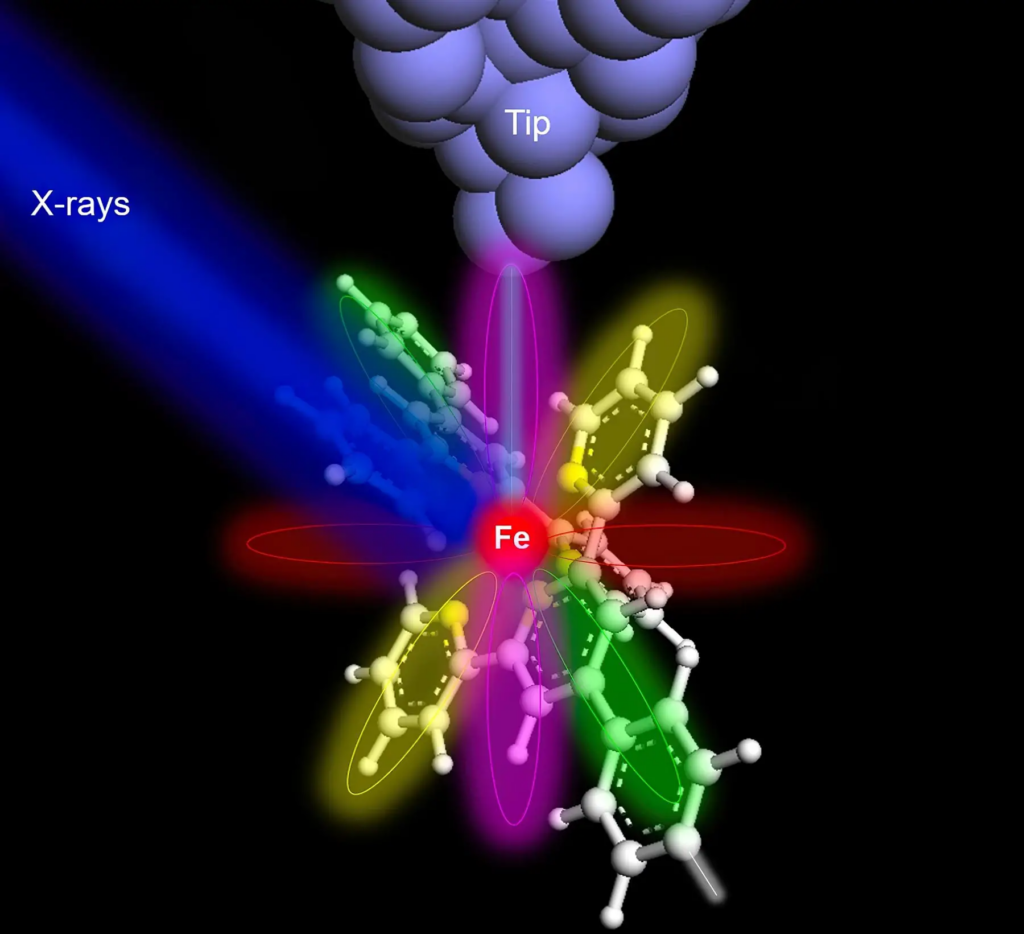
They created a specialized instrument that allowed them to examine atoms up close. This instrument is known as SX-STM, or synchrotron X-ray scanning tunneling microscopy. It measures the electrons agitated by the X-rays, which helps scientists determine which atom they are observing.
Atomic iron and terbium testing
To test their new method, the team placed a single iron atom and a single terbium atom within supramolecular assemblies, which are specialized clusters of atoms. These assemblies contained additional minute “building blocks.”
Not only were the researchers able to effectively image individual atoms using their X-ray technique, but they also obtained new insights into their chemical properties.
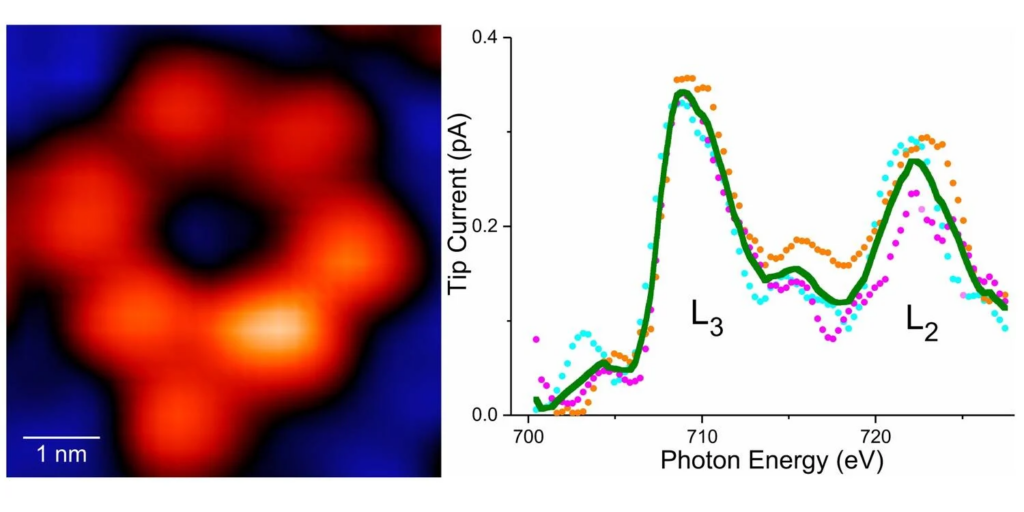
Professor Hla, the team’s leader, stated that they were able to distinguish the various chemical properties of individual atoms. Particularly, they discovered intriguing distinctions when comparing an iron atom and a terbium atom within their respective molecular substrates.
He added that the terbium atom remained relatively isolated and its chemical state did not endure significant alterations. On the other hand, the iron atom strongly interacted with its surroundings.
Tolulope Michael Ajayi, the paper’s first author and a Ph.D. student whose research significantly contributed to this discovery, stated that the method and concept utilized in this study have enabled significant advances in X-ray science and nanoscale investigations.
Ajayi explained further that the use of X-rays for the detection and characterization of individual atoms has the potential to revolutionize numerous fields of research.
